The Centre for the for the Cellular Microenvironment (CeMi) at the University of Glasgow brings together biomedical research themes in the areas of Biomaterials and Cell and Systems Biology, with state-of-the art facilities at the School of Engineering and at the Medical Veterinary and Life Science School.
Our labs focus on understanding the interactions between materials, proteins and cells to engineer and control cell behaviour and stem cell differentiation. The team benefits from strong links to the UK and international biomedical sciences and clinical medicine communities. We are home to 30+ PhD students and post-doctoral level researchers with projects ranging from basic material and biological science up to projects containing translational clinical components.
Stem cell engineering
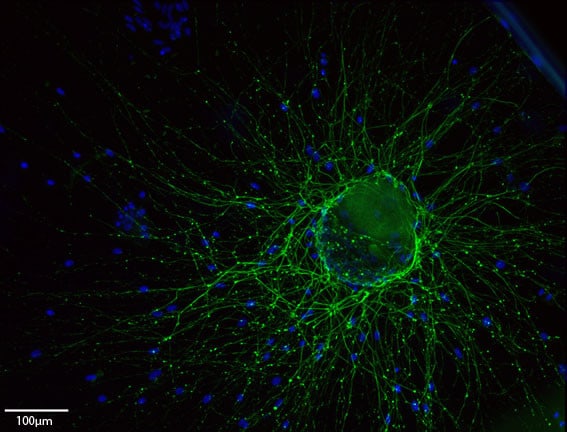
Cells produce and encounter rich extracellular matrix (ECM) environments around them, that help them perform their very diverse functions around the body. They use it as support and to regulate their dynamic behaviour. Engineering these cellular micro environments to promote regeneration or to model tissues in vitro, involves the design of complex systems that interact with cells in a variety of ways, mimicking what happens in living beings. We study and exploit interactions between material surfaces, synthetic biocompatible materials, natural and synthetic ECM molecules and growth factors (GFs) to understand and control cell behaviour and tissue repair.
We are pioneering work in several strategies to modulate cell behaviour and to direct stem cell fate:
- Nanovibrational stimulation to guide stem cell differentiation, and development of the ‘Nanokick Bioreactor’ to prime stem cells in vitro.
- Synthetic material-driven fibronectin fibrillogenesis, and it’s use to enhance presentation of GFs in different tissue engineering platforms.
- Introduced the concept of living biointerfaces to interact and study cell biology. We use genetically modified non-pathogenic bacteria as an in vitro functional and dynamic interface with stem cells.
Advanced functional biomaterials
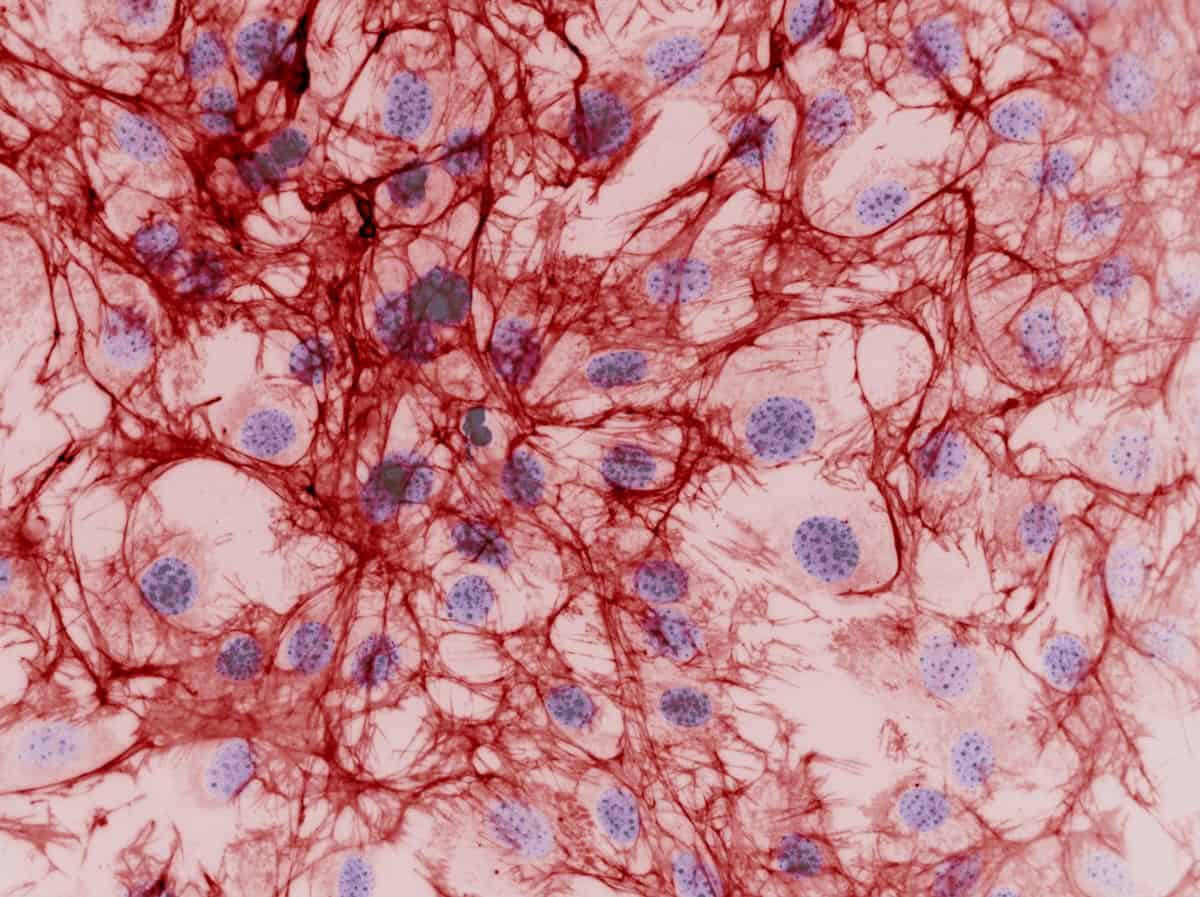
Advanced material-based systems are developed as part of tissue engineering strategies and stem cell–based regenerative therapies to promote cell survival, stem cell differentiation, better integration to host tissues and to deliver the stem cells themselves. In our labs we pursue new techniques for the fabrication of functional biomaterials using hybrid synthetic-natural hydrogels, 3D bioprinting, microfuidics encapsulation, novel polymer architectures and polymerisation technologies (e.g. low pressure plasma, atomic transfer radical polymerisation), surface modification and coating techniques, for applications in musculoskeletal, cardiovascular, neural, hematopoietic, vascular and wound healing systems.
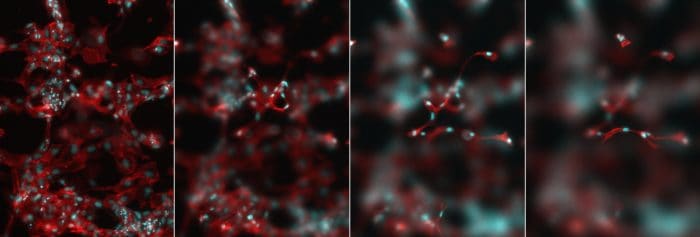
In vitro tissue models
Improved in vitro models enable the study of human tissues in health and disease (e.g. cancer, degenerative diseases, inflammatory diseases), the assessment of new drug delivery tools (e.g. functional biomaterials, encapsulation delivery technologies, new bioreactor designs and other cell culture systems, new microfluidic devices, etc.), and the cheaper and safer toxicity screening of new drug candidates. We develop robust extracellular matrix mimics, that provide the essential characteristics of a natural ECM in its ability to direct and control cell behaviour, yet with minimal complexity.
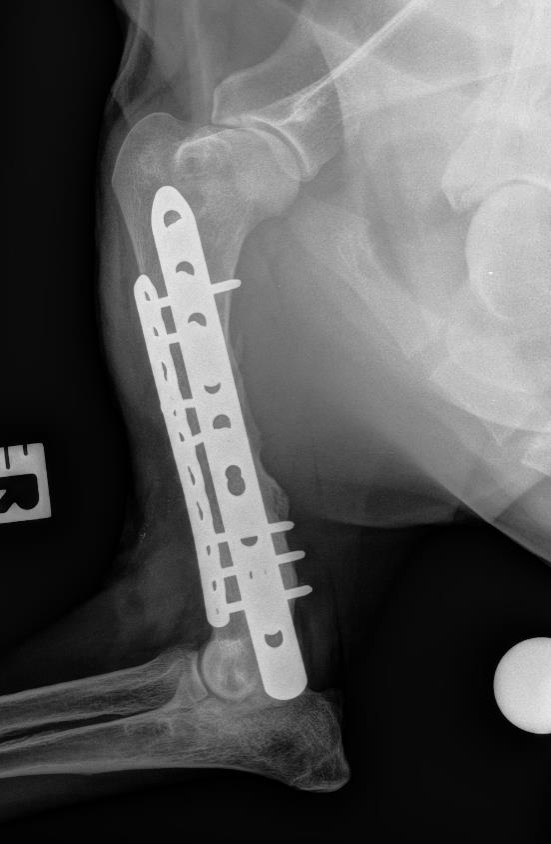
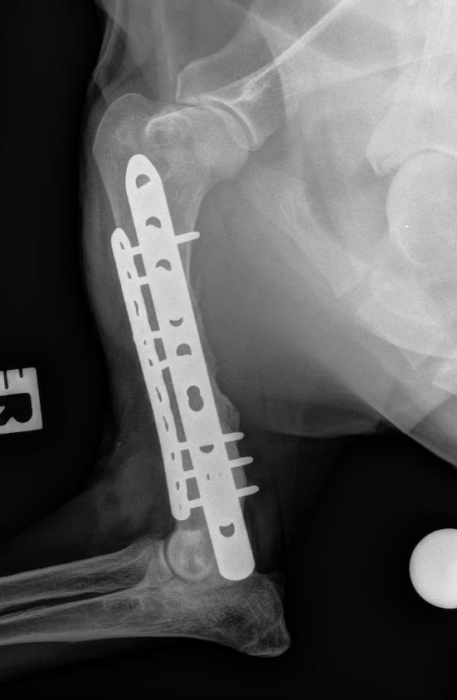
Bone tissue engineering
We work to translate therapeutic solutions that address clinical needs in bone regeneration. Our research relies in the use of acellular and cell based complementary strategies to guide stem cell differentiation into osteogenic precursors. Our work has progressed to preclinical models of non-union bone defects and we have planned to start clinical phases with a cell therapy in late 2020. Our core technologies for bone regeneration aim to:
- Develop in vitro implantable bone grafts, using our in house developed Nanokick bioreactors to promote stem cells to differentiate into osteoblasts.
- Develop safe and effective delivery systems for osteogenic and vasculogenic growth factors (e.g. BMP-2, BMP-7, VEGF) using material-driven fibronectin fibrillogenesis that enhance the presentation of growth factors.